NHC Patient-Focused Medical Product Development Webinar Series: Real World Case Examples
08/27/2020
By Kelsie Majercak, PharmD, BCACP, Research Intern
On August 13, the National Health Council (NHC) held a webinar to highlight how companies are moving from ad hoc to systematic patient engagement approaches and explore real-life case examples where patient input made a difference in drug development. The webinar featured:
- Keri Yale, MBA, Head, Patient Affairs & Engagement, Boehringer Ingelheim Pharmaceuticals, Inc.;
- Hilary Wilson, PhD, Director, Patient Affairs and Engagement, Boehringer Ingelheim Pharmaceuticals, Inc.; and
- Trisha Devlin, Director of Patient Value, Dermira a Wholly Owned Subsidiary of Eli Lilly.
Ms. Yale and Dr. Wilson described how Boehringer Ingelheim’s (BI’s) approach to patient engagement has evolved since 1998 when their HIV Community Affairs function was established. Through this evolution, BI identified three key pillars to advance patient engagement across BI: people, process, and progress.
- People: embedding patient engagement champions at every level of the organization, for example Patient and Professional Relations therapeutic area leads sit within pipeline and commercial project teams and serve as “strategic facilitators” of project/product specific patient engagement activities.
- Process: tools and systems to support patient engagement, for example a BI Guidance that outlines expectations for integration of patient insights in clinical development process.
- Progress: PE activities need to be tracked and evaluated to facilitate continuous improvement, for example a patient engagement quarterly report which describes patient engagement activities by disease areas and highlights best practices and examples.
In addition to describing this evolution, Ms. Yale and Dr. Wilson introduced participants to two examples of what patient engagement looks like in practice.
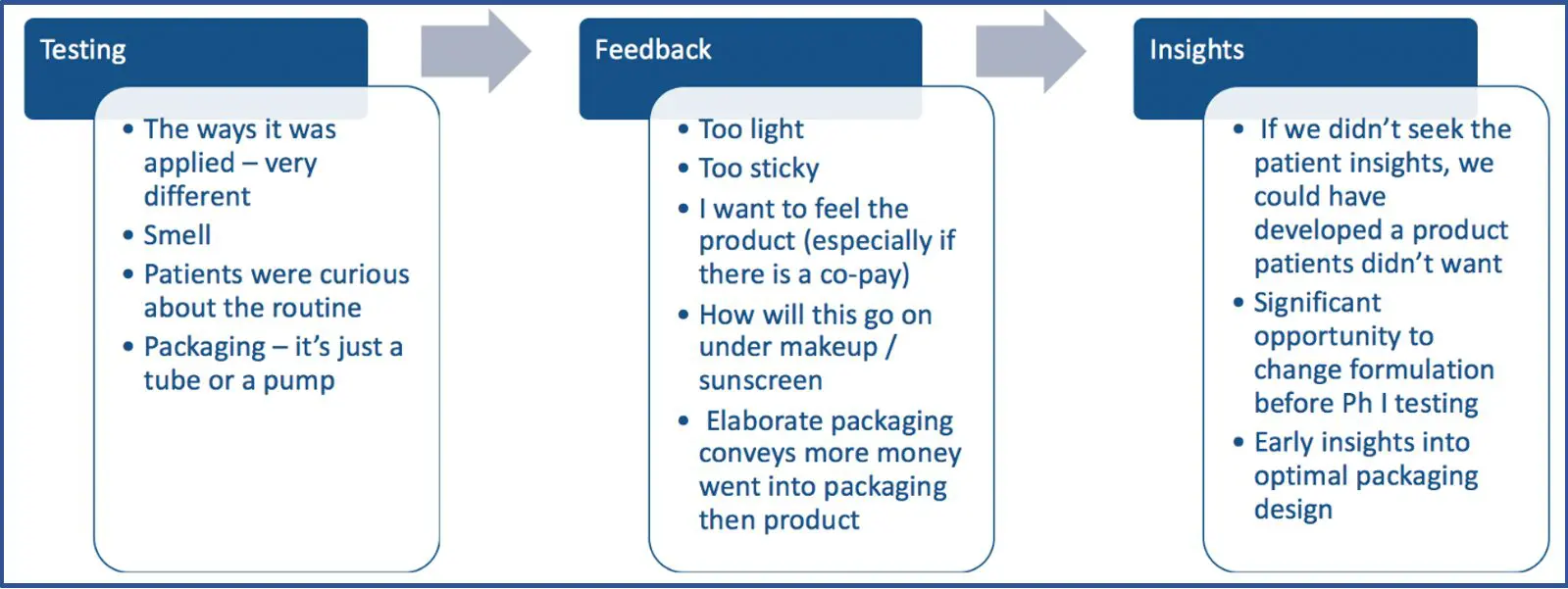
Next, Ms. Devlin described how the patient voice can be integrated across the drug development lifecycle. She shared a case example about how a pre-clinical team at Dermira was interested in engaging patients on a new formulation for a new therapy they were developing. They organized two focus groups of patients living with the disease to gather insights about current treatments, the potential new therapy, and packaging needs. Patient feedback is summarized in the figure below.
Ms. Devlin stressed the importance of engaging patients early, outlining the desired goal of each engagement, following up with patients when you say you are going to do so, and keeping metrics on ways patient insights changed developmental plans.
To listen to the complete webinar, click here. To read about other real-world case examples, visit our website here.

Join us Sept. 24 at 3 p.m. for our next Patient-Focused Medical Product Development (PFMPD) Webinar. Laurie Myers, MBA, Global Health Literacy Director, Merck, will discuss “Health Literacy Throughout Drug Development: Why it Matters to Pharma and to Patients.” Register Here.