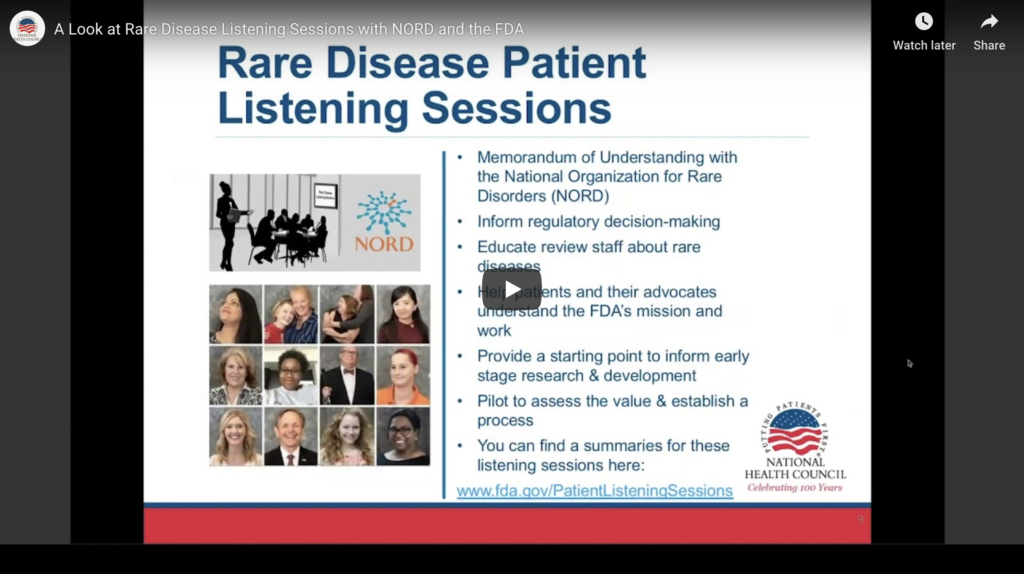
Patient-Focused Medical Product Development Webinar Series: A Look at Rare Disease Listening Sessions with NORD and the FDA
- Event Date: Jan. 31, 2020
- Event Time: 1:00 p.m.
- Event Location:
Our first webinar of 2020 will feature Debbie Drell, Director of Membership, at the National Organization for Rare Diseases (NORD), and Andrea Furia-Helms, Director, Patient Affairs Staff at Food and Drug Administration (FDA) to discuss their collaboration on Rare Disease Patient Listening Sessions. The listening sessions are facilitated by FDA Patient Affairs Staff and offer patients and caregivers an opportunity to speak directly to FDA staff about what it’s like to live with a specific rare disease. The sessions help FDA staff understand the patients’ perspective about their disease, needed treatment options, and meaningful outcomes.
Other Webinars

Optimizing Digital Health Trials by Engaging Patients and Sites
01/12/2021 2:00 pm, Virtual, ZoomRegister now for this webinar on Jan. 12 at 2 p.m. Speakers include, Virginia Nido, Global Head, Product Development Industry Collaborations, Genentech; and Cindy Geoghegan, Patient Advocate.
Read More
Health Literacy Throughout Drug Development: Why it Matters to Pharma and to Patients
09/24/2020 3:00 pm, Zoom Read More
Patient-Focused Medical Product Development: Real-World Case Examples
08/13/2020 2:00 pm, Zoom Read More